ACP-01: An Investigational Autologous Stem Cell Therapy for Ischemic Conditions
ACP-01: An Investigational Autologous Stem Cell Therapy for Ischemic Conditions
ACP-01 is an investigational autologous cell product derived from a patient’s own blood and studied in regulated clinical research for ischemic conditions involving impaired blood flow. Ongoing trials evaluate its role within vascular signaling environments under structured safety and regulatory oversight.
ACP-01
ACP-01 is an investigational therapy derived from a patient's own blood. It contains angiogenic precursor cells, specialized cells that play a role in blood vessel repair and circulation, and is being studied in regulated clinical research for conditions where blood flow to the heart, legs, or brain has become critically impaired.
ACP-01 is not an approved treatment. It is being evaluated in structured clinical research programs under independent ethical oversight and defined regulatory pathways. For patients who have exhausted standard options, it represents a science driven pathway worth understanding.
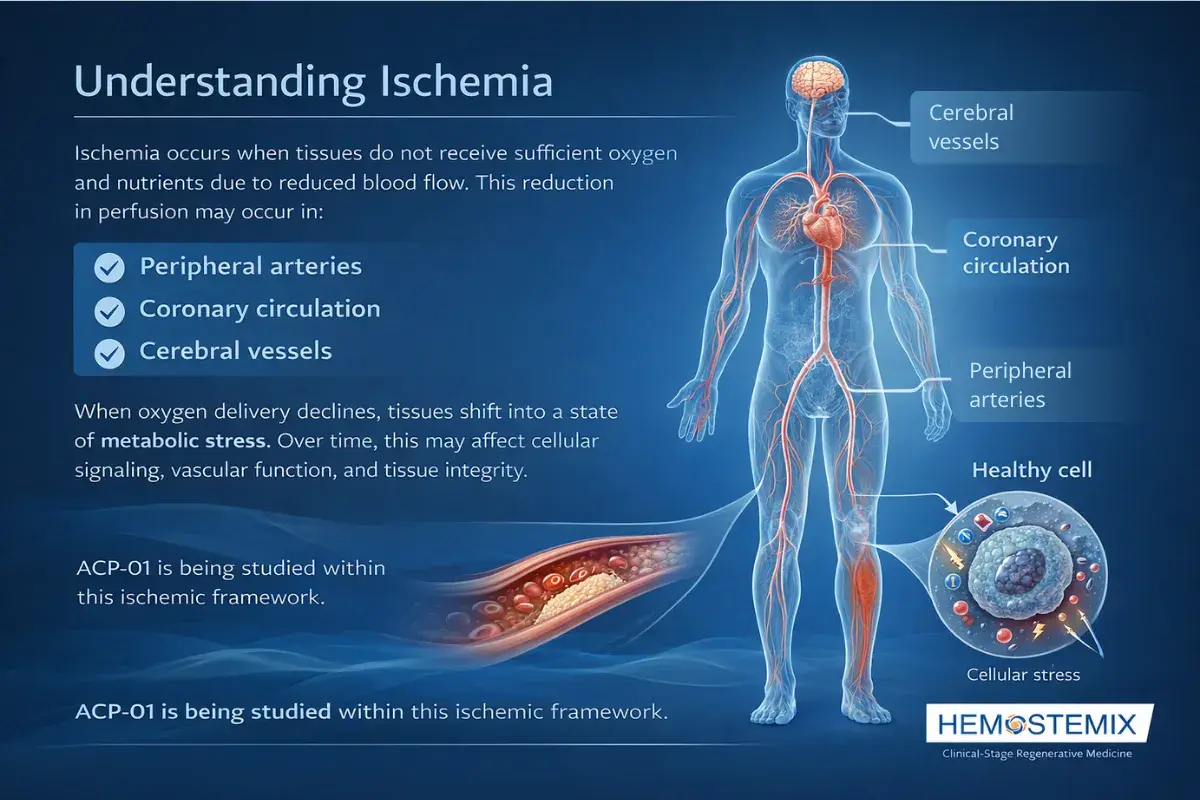
Understanding Ischemia
Ischemia is what happens when part of the body stops receiving enough oxygen-rich blood to function normally. It is not a single disease, it is a condition that can affect the heart, the legs, the brain, or all three simultaneously, because the underlying cause is almost always the same: blood vessels that have become too narrowed, damaged, or blocked to deliver what the tissue needs.
When oxygen delivery drops, the affected tissue becomes stressed. In the short term this produces symptoms like chest pain, leg cramping, or cognitive changes. Over time, if blood flow is not restored, the tissue weakens, scars, or begins to die.
This is the problem ACP-01 is being studied to address, not by bypassing or stenting a blocked artery, but by studying whether the body's own cells can be used to support vascular repair and improve circulation from within the tissue itself.
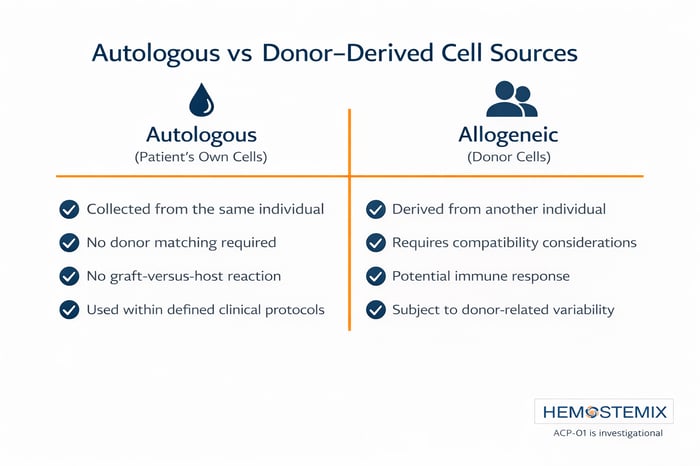
Why Autologous Matters
Why Autologous Matters
Autologous Cell Processing Platform
Autologous Cell Processing Platform
The process of creating ACP-01 begins and ends with the patient. There are no donor materials, no synthetic components, and no general anesthesia required at any stage.
The process involves four steps.
-
First, a standard blood draw is taken from the patient, the same procedure used for routine blood tests.
-
Second, the blood is processed in a controlled laboratory environment where angiogenic precursor cells are identified and concentrated.
-
Third, the prepared cells are quality checked under defined laboratory protocols before they are cleared for use.
-
Fourth, the cells are reintroduced into the area of ischemia using minimally invasive techniques under clinical supervision.
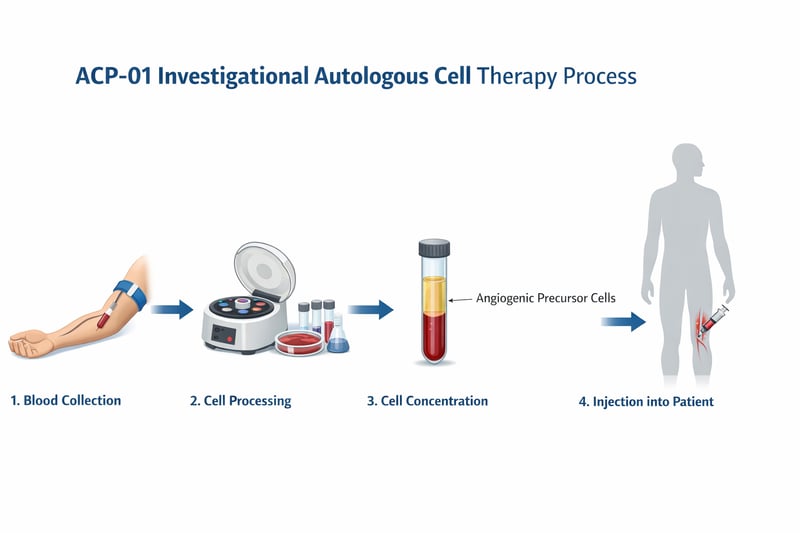
Recovery time varies by individual. The entire process is designed to be as straightforward as possible for the patient while maintaining the rigorous preparation standards required in a regulated research environment.
Mechanism of Action Under Investigation
ACP-01 contains angiogenic precursor cells, a type of cell found in the blood that plays a natural role in the body's vascular repair processes. Angiogenesis is the biological term for the formation of new blood vessels, and these cells are among the key players in that process.
When tissue is deprived of oxygen, the body attempts to compensate by signaling for new blood vessel growth to restore circulation to the affected area. In patients with advanced ischemic disease, this natural repair process is often insufficient on its own. The signaling is present but the response is too weak to overcome the degree of vascular damage that has occurred.
ACP-01 is being studied for its potential to support and amplify this natural process. By reintroducing concentrated angiogenic precursor cells directly into ischemic tissue, the research is evaluating whether those cells can interact with the existing vascular signaling environment in ways that support new vessel formation, improve microvascular function, and increase oxygen delivery to tissue that has been chronically starved of it.
The precise mechanisms and clinical implications remain under investigation. ACP-01 is investigational and no conclusions regarding therapeutic effect can be drawn outside of structured clinical study results.
Mechanism of Action Under Investigation
ACP-01 contains angiogenic precursor cells, a type of cell found in the blood that plays a natural role in the body's vascular repair processes. Angiogenesis is the biological term for the formation of new blood vessels, and these cells are among the key players in that process.
When tissue is deprived of oxygen, the body attempts to compensate by signaling for new blood vessel growth to restore circulation to the affected area. In patients with advanced ischemic disease, this natural repair process is often insufficient on its own. The signaling is present but the response is too weak to overcome the degree of vascular damage that has occurred.
ACP-01 is being studied for its potential to support and amplify this natural process. By reintroducing concentrated angiogenic precursor cells directly into ischemic tissue, the research is evaluating whether those cells can interact with the existing vascular signaling environment in ways that support new vessel formation, improve microvascular function, and increase oxygen delivery to tissue that has been chronically starved of it.
The precise mechanisms and clinical implications remain under investigation. ACP-01 is investigational and no conclusions regarding therapeutic effect can be drawn outside of structured clinical study results.

Safety Monitoring in Clinical Research
Every ACP-01 research program is built around patient safety first. This is not a regulatory formality - it is the foundation of how Hemostemix conducts research and the standard we hold ourselves to before any patient participates in any study.
Safety protections in every ACP-01 research program include clearly defined criteria that determine who is and is not eligible to participate, prespecified safety endpoints that are monitored throughout the study, independent Institutional Review Board review and approval before any research begins, and ongoing regulatory engagement under U.S. FDA oversight.
An Institutional Review Board is an independent ethics committee that reviews study design, patient protections, informed consent processes, and safety monitoring before human research may proceed. No study moves forward without their approval.
Clinical trials conducted under U.S. jurisdiction require Investigational New Drug clearance from the FDA prior to initiation. Hemostemix does not proceed without it.
Safety Monitoring in Clinical Research
Every ACP-01 research program is built around patient safety first. This is not a regulatory formality - it is the foundation of how Hemostemix conducts research and the standard we hold ourselves to before any patient participates in any study.
Safety protections in every ACP-01 research program include clearly defined criteria that determine who is and is not eligible to participate, prespecified safety endpoints that are monitored throughout the study, independent Institutional Review Board review and approval before any research begins, and ongoing regulatory engagement under U.S. FDA oversight.
An Institutional Review Board is an independent ethics committee that reviews study design, patient protections, informed consent processes, and safety monitoring before human research may proceed. No study moves forward without their approval.
Clinical trials conducted under U.S. jurisdiction require Investigational New Drug clearance from the FDA prior to initiation. Hemostemix does not proceed without it.
20 Years Of Clinical Research History
ACP-01 is not a new idea. It is the product of over two decades of structured clinical investigation in ischemic conditions, built on a foundation of peer reviewed published science and regulatory engagement.
Key milestones in the ACP-01 research program include the completion of a Phase II randomized clinical trial evaluating ACP-01 in patients with chronic limb threatening ischemia, with results published in the Journal of Biomedical Research and Environmental Sciences. A fourth cardiovascular study was published in Stem Cell Research and Therapy, adding to the peer reviewed body of evidence supporting continued investigation. Hemostemix completed a pre-Investigational New Drug meeting with the U.S. FDA and filed submission 1517 to advance ACP-01 across multiple ischemic indications simultaneously under a basket protocol framework. Institutional Review Board approvals have been secured for specific research protocols including angina and vascular dementia.
Across completed and ongoing studies, ACP-01 has been evaluated in structured clinical research programs involving hundreds of subjects across conditions including peripheral arterial disease, chronic limb threatening ischemia, cardiovascular ischemia and angina, and vascular dementia.
20 Years of Clinical Research History
ACP-01 is not a new idea. It is the product of over two decades of structured clinical investigation in ischemic conditions, built on a foundation of peer reviewed published science and regulatory engagement.
Key milestones in the ACP-01 research program include the completion of a Phase II randomized clinical trial evaluating ACP-01 in patients with chronic limb threatening ischemia, with results published in the Journal of Biomedical Research and Environmental Sciences. A fourth cardiovascular study was published in Stem Cell Research and Therapy, adding to the peer reviewed body of evidence supporting continued investigation. Hemostemix completed a pre-Investigational New Drug meeting with the U.S. FDA and filed submission 1517 to advance ACP-01 across multiple ischemic indications simultaneously under a basket protocol framework. Institutional Review Board approvals have been secured for specific research protocols including angina and vascular dementia.
Across completed and ongoing studies, ACP-01 has been evaluated in structured clinical research programs involving hundreds of subjects across conditions including peripheral arterial disease, chronic limb threatening ischemia, cardiovascular ischemia and angina, and vascular dementia.
Request A Clinical Research Consultation
Request A Clinical Research Consultation
If you have been diagnosed with an ischemic condition and want to understand whether ACP-01 research may be appropriate for your situation, the next step is a conversation with our clinical team.
Frequently Asked Questions
Frequently Asked Questions
What is ACP-01?
ACP-01 is an investigational autologous cell therapy derived from a patient's own blood. It contains angiogenic precursor cells that are being studied for their potential to support blood vessel repair and improve circulation in areas of the body affected by ischemic disease.
What does investigational mean?
Investigational means ACP-01 is still being studied in clinical research to evaluate safety and defined clinical endpoints. It has not been approved by the U.S. Food and Drug Administration for commercial use and is not available outside of structured research programs.
How is ACP-01 different from other cell therapies?
ACP-01 is autologous, meaning it comes from the patient's own blood. This eliminates the need for donor matching and removes the risk of immune rejection associated with donor derived cell therapies. The cells are recognized by the body as its own.
Is the procedure painful or difficult?
The process begins with a standard blood draw and ends with a minimally invasive reintroduction of the prepared cells. No general anesthesia is required. Most patients return to normal activities shortly after the procedure.
Is ACP-01 approved by the FDA?
No. ACP-01 is investigational and has not been approved by the U.S. Food and Drug Administration. All research is conducted under defined regulatory pathways and independent ethical oversight.
Who is eligible to participate in ACP-01 research?
Eligibility is determined by protocol specific inclusion and exclusion criteria that vary by condition and study design. The best way to find out whether you or a loved one may qualify is to request a clinical consultation with the Hemostemix team.
How do I find out if ACP-01 research is right for me?
Contact Hemostemix directly to request a clinical evaluation. Our team will review your medical history, current symptoms, and prior treatments to determine whether investigational research may be an appropriate next step for your situation.
Disclaimer

